A Fat is a Fat is a Fat...Right?
This week, the U.S. Food and Drug Administration (FDA) ruled that, over the next three years, trans fats must be removed from a wide range of processed foods. An artificially created fat developed to be solid at room temperature, trans fats raise the risk of heart disease.
With fats in the news, we thought it was a good time to dig a little deeper and discuss the various dietary fats. You may know that trans fats are bad, but do you know why? And do you know how they are different from saturated, monounsaturated and polyunsaturated fats?
Fats get a bad rap, primarily for their calorie content. At 9 calories per gram, it doesn’t take much fat to push our daily calorie intake too high. Only one tablespoon of olive oil contains 119 calories. But fats are essential for our bodies to function so you need to get some in your diet every day.
To better understand the differences in fats, we need a little chemistry lesson.
A dietary fat molecule is primarily composed of a long chain of carbon and hydrogen atoms. The carbon atoms may be linked by single or double bonds and the length of the chain can vary. The length of the chain and the number of carbon double bonds and their positions help define the type of fat and its role in the body.
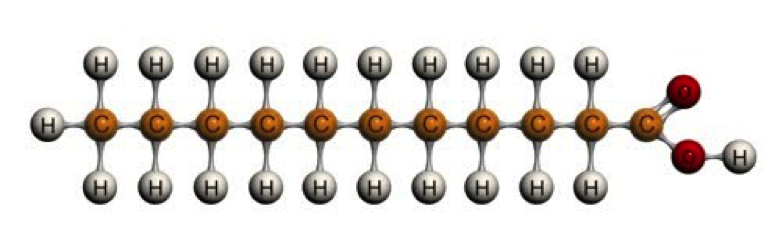
The simplest fat to describe is a saturated fat. It has a uniform-looking chain of carbon atoms all linked by single bonds. All other bonding sites along the chain are occupied (saturated) by hydrogen:

This structure makes saturated fatty acids solid at room temperature. Butter is a great example of a saturated fat.
Monounsaturated fats have one (mono) double carbon bond in the chain. Polyunsaturated fats have more than one (poly) double carbon bond. Both of these fats are liquid at room temperature. You’ll find monounsaturated fats in great abundance in olive oil while polyunsaturated fats are found in fish, nuts and seeds.
Here’s what a polyunsaturated fat might look like:

The position of the first double carbon bond defines the type. An omega 3 fat will have the first double bond at carbon atom #3, while an omega 6 fat will have its first double bond at carbon atom #6. Omega 3s are especially desirable as they have multiple health benefits especially for your cardiovascular system. Walnuts, flax and chia are all good sources of omega 3s.
You might have noticed that the hydrogen atoms surrounding a carbon double bond in poly and monounsaturated fats all fall on the same side of the carbon chain. This is the structure nature intended.
Trans fats, also referred to as hydrogenated and partially hydrogenated fats, are artificially modified to change that structure with hydrogen atoms found on opposite (trans) sides of the carbon. These fats are found primarily in margarines, processed and fast foods.

This seemingly minor variation in chemical structure completely changes the functionality of the fat. Rather than being liquid at room temperature, trans fats are solid, and rather than being vulnerable to rancidity, trans fats have a long shelf life.
In the 1970s, when saturated fats were thought to be major contributors to heart disease, the use of trans fats was encouraged, since they are technically an unsaturated substitute. Remember the commercials telling us to switch from butter (saturated) to the “healthier” choice margarine (trans)?
But it turns out trans fats are actually harmful to cell structure and promote heart disease to a greater extent than saturated fats ever could. In fact, the FDA estimates that eliminating these fats from the American food supply would translate into 20,000 fewer heart attacks and 7,000 fewer deaths per year.
Ironically, that old commercial was right after all: “It’s not nice to fool Mother Nature!”
There are a whole lot of details here, maybe more than you need. But here’s the takeaway:
- Fats are essential for our bodies to function properly, so we need some fat every day.
- Fat calories add up fast so limit your consumption.
- Avoid trans fats at all costs.
- Poly and monounsaturated fats, found only in plant-based foods, are beneficial for cardiovascular health.

Tested & Proven Results.
- Cardiologist formulated
- Supported by over 500 publications
- Clinically-proven, in a double-blind randomized trial with Mayo Clinic and The University of Manitoba
80% of participants lowered their cholesterol in just 30 days. With just two servings per day, Step One Foods offers a proven-effective way to naturally lower LDL (bad) cholesterol.
Get heart health tips and articles like this, delivered right to your email.
New articles every week.
You may also like...

Insulin Resistance, Prediabetes and Type 2 Diabetes. Part 4: Un-Doing It.

You don’t need to avoid foods with cholesterol…except for these



